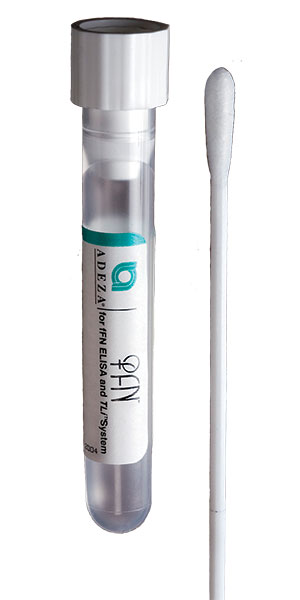
Fetal Fibronectin FFN
| Method(s) | Solid-Phase Immunochromatographic Assay |
|---|---|
| Specimen Required | |
| Collect | Fetal Fibronectin specific collection kit (swab in transport tube). See Specimen Collection Containers/Devices. |
| Transport | Transport specimens at 2-8ºC. Specimens should not be exposed to temperatures greater than 25ºC. |
| Remarks | 1. Special collection devices, with instructions, are provided by the laboratory (315-461-3008). 2. Take care not to contaminate with lubricants, soaps or disinfectants. 3. The specimen must be obtained prior to digital cervical examination, vaginal probe ultrasound examination, or collection of swabs for culture, as manipulation of the cervix may cause release of fFN. 4. Symptomatic Women: During a sterile speculum examination, lightly rotate the supplied swab across the posterior fornix for approximately 10 seconds to absorb cervicovaginal secretions. 5. Asymptomatic Women: During a sterile speculum examination, lightly rotate the supplied swab across the posterior fornix or the ectocervical region of the external cervical os for approximately 10 seconds to absorb cervicovaginal secretions. 6. Place swab in transport tube. Break the shaft (at the score). 7. Align the shaft with the tube cap and push down, tightly sealing the tube. |
| Stability | Ambient: 8 hours; Refrigerated: 3 days |
| Unacceptable Conditions | Specimens collected with wooden swabs or contaminated specimens. |
| Schedule | Daily |
| Billing Code | 1010277 |
| CPTCode | 82731 |
| Notes | The test should NOT be used in the following cases: |
| Preferred Specimen Collection Device(s) | |
| Reference Interval | |
|
In normal pregnancy, fFN is negative from 22-35 weeks. Positive results during this time are predictive of high-risk preterm delivery. fFN testing has been approved for risk determination in both symptomatic and asymptomatic patients. In symptomatic patients: a NEGATIVE fFN "rules out" preterm delivery (Less than 1% will deliver within the next 14 days), a POSITIVE fFN test allows intervention for those with the highest risk of delivery in the next 14 days. |
|