Maternal Serum Screen, Alpha Fetoprotein, hCG, Estriol, and Inhibin A (Quad) MSSQ
| Method(s) | Quantitative Chemiluminescent Immunoassay |
|---|---|
| Specimen Required | |
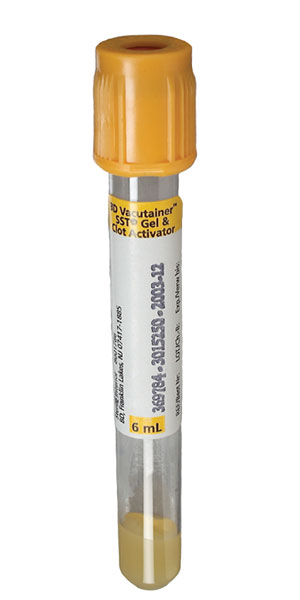
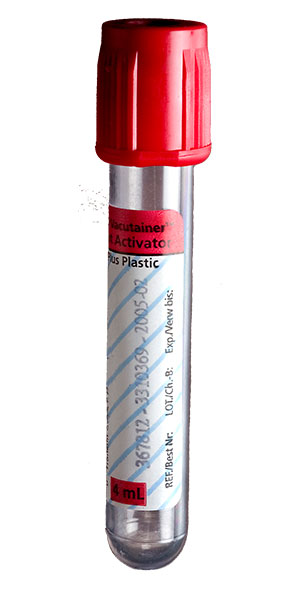
| Collect | One 5 mL gold (SST) or plain red top tube. |
| Transport | Separate serum from cells ASAP or within 2 hours of collection. Transfer 3 mL serum refrigerated to an ARUP Standard Transport Tube. (Min: 1 mL) |
| Remarks | Submit with Order: Patient's date of birth, current weight, due date, dating method (US, LMP), number of fetuses present, patient's race, if the patient was diabetic at the time of conception, if there is a known family history of neural tube defects, if the patient has had a previous pregnancy with a trisomy, if the patient is currently smoking, if the patient is taking valproic acid or carbamazepine (Tegretol), if this is a repeat sample, and the age of the egg donor if in vitro fertilization. |
| Stability | Collection to initiation of testing: After separation from cells: Ambient: 72 hours; Refrigerated: 2 weeks; Frozen: 1 year (avoid repeated freeze/thaw cycles). |
| Unacceptable Conditions | Plasma. Hemolyzed specimens. |
| Schedule | Daily |
| Billing Code | 5011815 |
| CPTCode | 81511 |
| Notes | See attached required requisition and patient consent. Patient Prep: Specimen must be drawn between 14 weeks, 0 days and 24 weeks, 6 days gestation. The recommended time for maternal serum screening is 16 to 18 weeks gestation. |
| Preferred Specimen Collection Device(s) | |